Dose-Response Relationships for Experimental Heterochrony in a Colonial Hydroid PDF
Preview Dose-Response Relationships for Experimental Heterochrony in a Colonial Hydroid
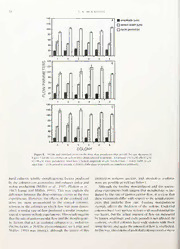
Reference:Hiol Bull 193:47-61.(August. IW7) Dose-Response Relationships for Experimental Heterochrony in a Colonial Hydroid NEIL W. BLACKSTONE DepartmentofBiologicalSciences, NorthernIllinois L'niversiiv. DeKalh, IllinoisM)l15 Abstract. Hydractiniid hydroids display a range of and uncoupling again have similar effects, triggering morphologicalvariation.Atoneendofthespectrum,the metabolicactivation(e.g., an increaseinoxygen uptake coloniesgrowinasheet-likeconfigurationwiththeirpol- andashiftinthemitochondrialredoxstateinthedirec- yps close together and short stolons. At the other ex- tion ofoxidation). In thecontextoftheoriessuggesting treme,thecolonieshavearunner-likeforminwhichthe the metaboliccontrol ofdevelopment,adirecteffect of polypsare fartherapart and connected by longstolons. feeding and uncoupling on colony development thus These patterns exemplify the heterochronic variation cannotberuledout.Further,theremaybeaninteraction found in many colonial animals and correspond to between flowrateand metabolism,sincegastrovascular changes in the timingofthe production ofpolyps and flow distributes food throughout the colony, and since stolon tipsrelativeto ratesofstolongrowthandcolony such substrate affects metabolic state. Both within-col- maturation. Experimental studiesofclonal replicatesof onyflowrateand metabolism mayaffect heterochronic a Podocoryne carnea colony demonstrated a dose-re- variationinthesehydroids.andmethodsappropriateto sponse relationship between these heterochronic traits distinguishthesetwoeffectsarediscussed. andwithin-colonygastrovascularflowtoperipheralsto- lons. A dose-response relationship was found whether Introduction flowwasperturbedbymanipulatingtheamountoffood consumed by the colonies or by treating the colonies Evolutionary changes in the timing ofdevelopment with2,4-dinitrophenol,anuncouplerofoxidativephos- underlie much ofanimal diversity (e.g.. Gould. 1977; phorylation. In colonies in which flow was highly per- Alberch el a/.. 1979; Bonner. 1982; McKinney, 1988; turbed by either treatment, a similar rate offlow pro- Wake el al.. 1991; Hall, 1992). Historically, studies of ducedasimilarmorphologicalresponse.Thesedatasup- heterochrony have focused on describing patterns of portthehypothesisofacausalrelationshipbetweenflow comparative embryology and morphology (Gould, rateandheterochronicvariation.Nevertheless,flowwas 1977),andtoalargedegreethisdescriptiveandcorrela- diminished by two clearly different mechanisms. Feed- tional tradition still persists(see Raft"and Wray, 1989). ingmanipulation altered flow relativetothesizeofthe Thus, relatively little isknownabout theactual mecha- stolon byalteringstolonthickness,withoutaffectingthe nismsthatgovernheterochronicvariation.Aremorpho- absolutequantityofflow. Uncouplingwith dinitrophe- logical and life-history heterochronies direct conse- noldiminishedtheabsolutequantityofflow,butdidnot quences ofgenetic and molecular heterochronies? Do affect the size ofthe stolon. A plausible assumption is physiological,developmental,andmetabolicfactorsme- that feeding manipulation affects the resistance ofthe diate heterochronic variation? Can unrelated genetic stolontissuetoflow,orthefluidabsorptionofthistissue, changesproducesimilarheterochroniesbyaffectingthe orboth; whereas uncoupling affectsthe amount ofen- same epigenetic process? Investigations ofsuch ques- ergy available to drive the flow. At the level ofcellular tionsarecrucialifstudiesofheterochronyaretoprogress metabolism, on the other hand, feeding manipulation beyond largely descriptive studiesofmorphology (Raff andWray, 1989). To address these sorts ofquestions, whole-organism Received19September1996:accepted7May 1997. experimental manipulations have become increasingly 47 48 N. W. BLACKSTONE common in studies of evolutionary morphology (cf. sexual reproduction) and paedomorphic (i.e.. "child- KettersonandNolan, 1992;Sinervo >//., 1992;Sinervo shaped")adultmorphologies. andBasalo, 1996).andthisisparticularlytrueinstudies Hydractiniidhydroidsillustratethesegeneralpatterns: oftheevolutionofdevelopment(e.g..AlberchandGale. speciesofHydractiniaaretypicallysheet-like,whilespe- 1985: Stebbins and Basile, 1986; Meyer. 1987; Miiller. ciesofPodocorynearetypicallyrunner-like(Blackstone 1991; DeSalle and Carew, 1992; Blackstone and Buss. and Buss, 1991; Blackstone. 1996). The morphological 1992. 1993:Dudgeonand Buss. 1996).Thecentralmo- aspectsofthisvariation derivefrom the higherratesof tivationfortheselatterstudiesisarticulatedbyStebbins polypandstolontipformation,relativetoratesofstolon and Basile(1986) in theirdefinition ofphyletic pheno- growth and colony maturation, in Hydractinia com- copies: "We propose this term for changes in form or paredtoPi>dncoryne. Thisdifferenceisparticularlypro- physiological response that mimic the normal form or nouncedatthetimeoftheformationofthestolonalmat reactionofarelatedphenotype,particularlyonebelong- in llydructiiiiii. In addition, the relative rates ofpolyp ingtoadifferenttaxon. Usingthem, investigatorshave andstolonproductionshowan inversecorrelationwith obtainedcluestothedevelopmental basisofevolution- ratesofgastrovascularfluidflowtoperipheralstolontips. arychange,andoccasionallytothenatureandactionof ComparedtocoloniesofPodocorynecornea,maturecol- the genes involved." Experimental aspects ofthis ap- onies of llYilnictiniu symbiolongicarpus exhibit a low proach can be particularly effective in the context of rateofflowtoperipheralstolons,particularlysubsequent studies ofheterochrony (e.g., Meyer, 1987). Neverthe- to the formation ofthe stolonal mat (Blackstone and less,todefinitivelyinvestigatethebasisforheterochrony. Buss, 1992;Blackstone. 1996). such experiments should include notjust gene dosage Experimental studies of heterochrony in these hy- studies(e.g., DeSalleandCarew, 1992),butalsomanip- droidshavedemonstratedthatthebetween-speciespat- ulations of physiological and developmental mecha- terncanbemimickedbyexperimental manipulationof nisms against a uniform genetic background. Further, coloniesofasinglespecies(Blackstoneand Buss, 1992. once an experimental basis for heterochrony has been 1993). Putatively,thesehydroidsincursubstantialener- demonstrated, the causal basis of such a relationship geticcostsincirculatingthegastrovascularfluidthrough- shouldbefurtherelucidatedbyestablishingtheunderly- outthecolony. Application of2,4-dinitrophenoltocol- ingbiologicalgradientordose-responsecurve(e.g.. Hill, onies of Podocoryne carnea results in a condition of 1965;Weed, 1988). "loose-coupling" of oxidative phosphorylation. a de- Asmodelsystemsforexperimentalstudiesoftheevo- creaseintheenergyavailableforgeneratinggastrovascu- lution of development, clonal organisms (e.g., many larflow,andaconsequentdiminishingoftherateofflow fungi,herbaceousplants,andcolonialinvertebrates)are to peripheral stolons. Correlated with this diminished particularlyappropriate. In such organisms,thegrowth flowarechangesthat parallel patternsofheterochrony; and development of the colony are inseparable, and therateofproductionofpolypsandstolontipsincreases thereisabroadchronologicalwindowinwhichmanipu- relativetotheratesofstolongrowthandcolonymatura- lations ofdevelopment are possible; moreover, geneti- tion, and peramorphic("shapesbeyond") forms result. cally identical clonal replicatescan be used in theseex- Alternatively,gastrovascularflowcanbediminished by periments(Bussand Blackstone, 1991). In manyclonal increasingthenumberoftimesacolonyisfed(e.g..from groups, the morphologycan beidealizedascomprising 3to6timesperweek),possiblybecauseahigherrateof feeding and reproductive entities, here termed polyps, feedingincreaseseithertheviscosityofthegastrovascu- which are interconnected by vascularstolons. Runner- larfluid ortheresistanceto flow ofthestolonal tissues, like forms(cf.. "guerrilla" ofHarper, 1985)showwidely orboth. Feedingmanipulation, liketreatmentwith un- spacedpolypsandlongstolonalconnections,whilesheet- couplers, resultsinchangesthatparallelpatternsofhet- like forms(cf. "phalanx" ofHarper, 1985)showclosely erochrony; again, the rate ofproduction ofpolypsand packed polyps with short stolonal connections. These stolontipsincreasesrelativetoratesofstolongrowthand differentmorphologicalpatternscorrespondtochangesin colonymaturation(seeBraverman, 1974).Althoughin- thetimingoftheproductionofpolypsandstolontipsrel- creasedfeedingproducesasurfeitofnutrientsandseems ativetotheratesofstolongrowthandcolonymaturation: in manywaystheoppositeoftheenergy-poorstatepro- high rates ofproduction yield sheets, whereas low rates duced by uncoupling, its effect on colony physiology yieldrunners.Further,thesedistinctivemorphologiescor- (i.e.. flow rate)issimilar. Incombination,thebetween- relatewithavarietyoflife-historytraits;runner-likeforms species data (Blackstone and Buss, 1992; Blackstone, tendtogrowquickly,reproduceearly,anddispersewidely 1996)and theexperimental manipulations(Blackstone as compared to sheet-like forms (e.g., Jackson, 1979; andBuss, 1992, 1993)suggestthatflowrateisaprincipal Harper, 1985). In theterminologyofheterochrony, run- mechanism underlying heterochronic alterations of ner-like forms often exhibit progenesis (i.e.. precocious thesehydroidcolonies. HETEROCHRONY DOSE-RESPONSE IN HVDRO1DS 49 Nevertheless,theseinterpretationsarecomplicatedby plications,andthemethodsappropriatefortheirresolu- thesimilaritiespotentiallyinducedbyfeedingmanipula- tion, are discussed. At this time, it seems possible that tion and uncoupling treatments at the level ofcellular both colony physiology(i.e.. flow rate)and metabolism metabolism (e.g.. Chance et al., 1963; Heytler, 1981: maymediatethegeneticaspectsofheterochronyinthese Chance. 1991).Briefly,feedingtriggersmetabolicactiva- hydroids. tion, which has features (e.g.. increased oxygen con- sumption, shift ofthe mitochondria! redox state in the MaterialsandMethods directionofoxidation)thatcanbemimickedbyuncou- Studyspecies pling.Inthislattercase,oxidationofsubstrateis"uncou- pled" from energy conversion, so metabolic activation Podocoryne carnca exhibits traits typical of runner- does not lead to increased ATPformation. Treatments likecolonialanimals(Blackstone, 1996). Colonydevel- involving feeding manipulation and uncoupling may opment begins with the metamorphosis ofthe planula thussharesomecharacteristicsatthelevelofmitochon- larva into a primary polyp. Runner-like stolonsextend drial metabolism, but diverge at the level ofATP pro- from the primary polyp. Stolons encase fluid-filled ca- duction. Further,thefeaturesofmetabolicactivation in nalsthatarecontinuouswiththegastrovascularcavityof experiments with uncouplers are often sensitive to the thepolyp.Incrosssection,stolonsconsistofafluid-filled nature ofthe particular uncoupler used, its concentra- lumenencasedbyendoderm,ectoderm,andarigidperi- tion,andotheraspectsoftheexperimentalprotocols. derm.Gastrovascularfluidcirculatesinthelumenofthe Giventheseconsiderations,furtherinvestigationsinto stolonsandcarriesfoodand possibly other metabolites the relationship between gastrovascular flow rate and fromthefeedingpolyptootherpartsofthecolony:con- heterochronic variation in these hydroids are needed. tractionsofthemuscularpolyppropelthegastrovascular Here, two series ofexperiments are reported. The first fluid (Schierwater et al., 1992). As the lumen fills and seriesfocusedoncausalcriteria: ifadose-responserela- emptiesinresponsetocontractionsofthepolyps,theen- tionshipexistsbetweenflowrateandheterochronicvari- dodermalandectodermaltissuelayersofthestolon ex- ation,acausallinkbetweenthesevariablesgainsconsid- pandandcontractaswell.Therigidperiderm,however, erablesupport(e.g..Hill, 1965:Weed, 1988).Hence,two remainsfixedandsetsthemaximumdiameterforstolo- dose-responseexperimentswerecarriedoutwith clonal nalexpansion.Incrosssection,thestolon(andlumen)is replicatesofthesameP. carncacolony.Thefirstexperi- notcylindrical but moreclosely resemblesa halfellipse ment examined dose-response in feeding experiments. withthe"flat"surfaceadjacenttothesubstratum. Dur- Althoughtheactualexperimentaltreatmentisthenum- ingtissueexpansionandcontractioninresponsetoflow, beroffeedingsperweek,gastrovascularflowrate(thehy- thelumencrosssectionmaychangeshape,therebycom- potheticalphysiologicaldose)isconsideredthepredictor plicating the physical biology offluid flow (see Black- variable;morphologicaldevelopmentrelativetothetim- stone, 1996;VanWinkleandBlackstone, 1997). ingofthesexual(medusoid)phaseofthelifecycleiscon- Colony development from a primary polyp can be sideredtheresponsevariable.Thesecondexperimentex- mimicked by surgically explanting 1-2 polyps from a amined dose-response in uncoupling experiments. colonyontoa newsurface. In both cases, P. carncade- Again,gastrovascularflowrate(thehypotheticalphysio- velops by lineal extension ofthe stolons, initiation of logical dose) is considered the predictor variable, and newstolonaltips,anditerationoffeedingpolypsonthe morphologicaldevelopmentrelativetothetimingofthe stolons,andformsaloosenetworkofpolypsandstolons sexual (medusoid) phase ofthe life cycle is considered typical ofmany runner-like forms. Once the available theoutcomevariable. Bothexperimentssupportthehy- substratumiscovered,P.carneacoloniesincreasepolyp pothesisofadose-responserelationshipbetweengastro- and stolonal tip formation, producing a more closely vascular flow and heterochronic variation in these hy- knitnetworkofstolonsandultimatelyinitiatingthesex- droids. ual(medusoid)phaseofthelifecycle. Whengenetically Thesecondseriesofexperimentsdirectlyinvestigated identicalclonalexplantsofthesamecolonyareused,the metaboliccharacteristicsofcoloniessubjectedtofeeding developmentoftreatedcoloniescanbemanipulatedrel- manipulationanduncoupling;oxygenuptakeandmito- ative to control colonies, producing more sheet-like P. chondria!redoxstateweremeasuredinclonalreplicates carnea and thus providing an experimental system to ofthesameP. carneacolony. The resultssuggest some generally examine heterochronic variation in runners parallelsbetweenthesetreatmentsatthelevelofcellular andsheets(Fig. 1). metabolism,althoughtheextenttowhichsuch features can directly affect colony development remains unex- Productionofcoloniesandcultureconditions plored.However,theseresultscomplicatetheinterpreta- Coloniesonhermitcrabshellswerecollectedfromthe tion ofthedose-response relationships, and thesecom- Yale Peabody Museum Field Station in Connectku . 50 N. W. BLACK.STONE FigureI. Background-subtractedimagesofseveraltreatedandcontrolcoloniesofPodocorynecarnea: (A)treatedwith 30nMdinitrophenol:(B)dinitrophenolcontrol;(C)fed6timesperweek;(D)feeding control.Thegeneticallyidenticalcoloniesencrust 15-mmdiameterglasscoverslipsandwereimagedat thetimeoftheinitiationofmedusaproduction(datafromtheseimagesareincludedinFigs.3and6).In eachimage,polypsarebrightandcircular,stolonsaredarkerandweb-like,andthesubstratumappears black.Bothincreasedfeedinganduncouplingincreasethenumbersofpolypsandencrustingstolonsatthis developmentallandmark;suchcoloniescanbeconsideredperamorphic;/c,theyexhibit"shapesbeyond" thenormaldevelopmentalpathways. Clonal replicatesofoneP. carneacolonyweremadeby ulationexperiments,colonieswerefedtorepletionwith explanting 1-2polypsandtheirconnectingstolonsonto brine shrimp nauplii 3days per week. Analysis has round glass cover slips. For measures offlow rate and shown that, with similarculture conditions, "random" morphology, 15-mm diametercoverslipsare ideal, but statistical effects (e.g.. time effects, tank effects, rack formeasuresofoxygen uptakeand redox state. 12-mm effects;seeSokalandRohlf, 1981)arenegligible(Black- diametercoverslipsarenecessary.Aftertheexplantsat- stoneandBuss, 1991). tachedandbegantogrow,alltheoriginalexplanttissue wasremoved from thecoverslip. Inallexperimentsex- Colonyimageanalysisandmorphometrics cept the ones measuring redox state, colonies were effectivelyconfinedtoonesideofthecoverslipsbycut- Coloniesweremeasuredusingimageanalysistechnol- tingback encrusting stolons from the reverse side on a ogy(seeRohlfand Bookstein, 1990). Briefly,ahigh-res- dailybasis.Coverslipsweresuspendedin floatingracks olution MTI CCD-72 cameraattached toa macro lens andgrown in 120-literaquariacontainingReefCrystals wasusedtoprojectanimageofeachcolonyontoacolor artificial seawater (salinity == 35%o) with temperature monitor interfaced with a PC compatible microcom- controlto20.5 0.5C,undergravelfiltration,and50% puter(pentium/90MHz CPU, 32 Mb RAM)equipped waterchanges weekly. Ammonia, nitrites, and nitrates withanOverlayFrameGrabberboard(640x480pixels weremaintainedbelowdetectablelevels(AquariumSys- with 12-bit depth per pixel). OPTIMAS software was temstestkits).Withtheexceptionofthefeedingmanip- usedtoacquirebackground-subtractedimagesofthecol- HETEROCHRONV DOSE-RESPONSE IN HYDROIDS 51 onieswithilluminationappropriatetoproducethreedis- flowingseawaterwasadjustedwithathermoelectricde- tinctluminancethresholds:thepolyps(lightest),thesto- vicetomaintainaconstantchambertemperature(20.5 lons(intermediate), and theemptycoverslip(darkest). 0.3C; chamber temperature was monitored with a Usingthesethresholds,thesoftwareidentifiedandmea- YSI cuvette thermometer with a flexible probe). Colo- suredtheareasoftheempty(i.e.,unencrusted)coverslip nieswereviewedonan inverted lightmicroscope(Zeiss and of the individual polyps (Blackstone and Buss, Axiovert 135),witha40X Plan-Neoobjective in differ- 1992). Classification macros were used to identify and ential interference contrast. Three primary stolon tips excludeareasofcoverslipoutsidetheedgeofthecolony. from each colony were videotaped with the MTI CCD Thetotalcolonyareaandperimeterwerealsomeasured. camerafor 10minuteseach. Data fileswereanalyzed using PC-SASsoftware. For Gastrovascular flow must be reversed in each distal, eachcolonyateach measurementtime,thetotalareaof "dead-end"tip.Stolontipsfillasfluidenters;thevelocity polyps and the total area ofempty, unencrusted cover ofthe fluid then decreases to zero (Fig. 2). Tips then slipenclosedwithinthecolonywereexpressedasafrac- empty,andthefluid velocityagaindecreasestozero. In tionofthetotal area(notethatthetotal areaofstolons theregionofthestolon immediatelybehindthetip,the can be calculated as 1 minus this combined fraction). differencebetweenthewidthofthestolonlumenwhenit Polyp area is clearly a measure ofpolyp development; isatamaximum(andfluidvelocityiszero)andwhenit empty, unencrusted inner area is largely a measure of isata minimum (and velocity isagain zero)providesa stolondevelopment. Althoughpolypscanshieldempty, measureoftherateofgastrovascularflow, ifthisdiffer- unencrusted area from observation and measurement, ence is measured over time. These width or diameter in practicethisisaminorsourceoferrorbecausestolon measuresare taken at the base ofthe lumen. With the development is generally most extensive at the base of imageanalysissystemconnectedtotheVCR,thediam- thepolyps.Thisisparticularlytrueatthetimeofinitia- eter of the stolon lumen was measured at a point tionofthesexual(medusoid)phaseofthelifecycleand ~250/jm behindthetipitself. Inthisregion ofthesto- subsequently. Thus, at the times in development when lon,gastrovascular fluid velocitygoesto zero asthelu- morphology was measured (see below), polyparea and men diameterapproachesitsmaximum and minimum unencrusted inner area behave as largely independent (thus velocity itselfneed not be measured). Lumen di- measuresoftwodifferentaspectsofdevelopment. Mor- ameterwasmeasuredwhenthestolonwasfullandwhen phologicaldifferencesbetweenexperimental treatments itwasempty,for3consecutive,butnonoverlapping,cy- wereassessedbytwomethods:first,usingananalysisof cles.Foreachcycle,thenetlumenamplitude,thatis,the variance with the outcome equal to the natural loga- differencebetweenthemaximumand minimumlumen rithmoftheratiooftotalpolypareadividedbytotalun- diameters, was calculated. Periderm-to-periderm total encrustedinnerarea:second,usingamultivariateanaly- stolonwidth(whichisinvariantthroughoutthecontrac- sisofvariancewithtwooutcomes,thenaturallogarithm tioncycle)andtheperiod(inseconds)ofeachcyclewere ofthe ratio oftotal polyp area divided by total colony alsomeasured(Fig.2). areaandthenaturallogarithmoftheratiooftotalunen- Statistical analysis focused on the three measured crustedinnerareadividedbytotalcolonyarea.(Natural outcomes: lumen amplitude, cycle period, and stolon logarithmswereusedtobettermeettheassumptionsof width.Theinterpretationofthesemeasuresintermsof parametric statistics.) Both analyses ask essentially the thevolumetricrateofgastrovascularflowhasbeendis- samequestionsin slightly differentways, and bothgive cussed in detail elsewhere (Blackstone. 1996). Briefly, very similarresults. To avoid redundancy, only the re- these measures can be combined into a biologically sults ofthe MANOVA's are reported here. Additional meaningfulmeasureofgastrovascularflowrate:lumen analyses usingother measuresofmorphology (e.g., the amplitude divided by cycle period and stolon width ratiooftotal polypareato mean innerarea; see Black- (micrometersoflumendiameterpertotal micrometers stone and Buss. 1992, 1993) provide very similar be- ofstolonwidthpersecond). Biologically,thisratemea- tween-treatmenteffects. sureilluminatestherateofsupplyoffoodtothetissues ofthe stolon tip. Both this rate measure and the indi- I'ideomicroscopicmeasuresofperipheral ovifdpuaarlafmleotwripcarstaamteistteircss(gseeneerSaolklaylmaenedtRtohhelfa,ss1u9m8p1t).ioTnos gastrovascularflow comparetreatments, a nested analysisofvariancewas Gastrovascular flow reaches a maximum 2-8h after used with cycles nested within stolons, stolons nested feeding(Schierwaterelal., 1992);allofthesestudieswere withincolonies,andcoloniesnestedwithintreatments. carriedout3-5 hafterfeeding.Thecolonywasplacedin In somecases, near-significanteffectsthatmaybebio- aflow-through chamberwitha#1 coverslipasitsbase logically relevant are discussed (for justification, see (Warner Instruments). The temperature of the in- Rothman, 1986). 52 N. W. BLACKSIONI 40- A;UTMMAENXWMIDUTMH epncrtiondretremrm I 35- lumenwidth stolonwidth 30 lumencross-section UQJ 25 20 15- 10 \LUMENWIDTH ATMINIMUM 50 100 140 190 240 SECONDS Figure2. Thecontractionandexpansionofthestolonlumenasittillsandemptieslargelyinresponse ttoragnassvterrosveassceucltiaornHsowa,nd.Acmrpolsistsuedcetifoonrseoafcthhceysctloelcoonrtriepswpiotnhdsthteotlhuemdeinamaettmearxoifmtahlelaunmdenm.inSicmhaelmawtidatsh.hoIwn theregionofthestolonimmediatelybehindthetip.thedifferencebetweenthewidthofthestolonlumen whenitisatamaximum(andfluidvelocityiszero)andwhenitisataminimum(andvelocityisagain zero)providesameasureoftherateofgastrovascularflow,ifthisdifferenceismeasuredovertime(modified fromBlackstone Feedingmanipulationexperiments formation (Blackstone and Buss. 1992. I993). Dinitro- Eighteennewlyexplantedclonalreplicateson 15-mm cpohnetnaoilnitnrgea5t0memnltowfatshecaarprpireodproiuatteinsoglluatsisonp.etEriacdhischoels- cover slips were randomly divided into three groups. onywaskeptinasinglepetridish. Disheswerearranged Eachgroup received adifferent numberoffeedingsper on threetrays(one foreachtreatment) in an incubator week (2X, 3X [control], and 6X). The morphology of (20.5C). To randomize position and shelfeffects, each each colony was measured at the time the substratum colony'sdishwasshiftedbyoneposition eachday,and becamecovered,whenmedusaproductionwasinitiated, trays were moved to a different shelfeach day. Petri andwhenthefirst medusaewerereleased. Immediately disheswerechangeddaily.Whencolonieswerenotbeing precedingthefirst measure,gastrovascularflowtothree treatedwithdinitrophenol,theywerekeptintheaquaria stolon tips percolony was measured. The initiation of in the normal fashion. Colony morphologyand gastro- medusaproductionwasdefinedasthedayafterthefirst vascularflow ratewere measured at the same develop- medusa buds became visible on gonozooids ofthe col- mentallandmarksasinthefeedingmanipulationexper- ony.Thetimeofthefirstreleaseofmedusaewasdefined iments. as the day after maturing medusae (e.g.. tentacles ex- tended and swimmingbell fully extended and contrac- tile)werefirstobservedongonozooidsofthecolony. Dose-responserelationships In evaluating dose-response relationships between L'ncouplingexperiments physiologicalparameters(gastrovascularflowrateinthis Eighteennewlyexplantedclonalreplicateson 15-mm case) and a morphological outcome, several considera- cover slips were randomly divided into three groups. tionsarise. A considerabledifference isexpected in the Eachgroupwastreatedwithadifferentconcentrationof rangeofvaluesbetweenaphysiologicaldoseandamor- 2. 4-dinitrophenol in seawater(0pM [control], \5nAl, phological response. A physiological measure is rela- and30^Af)for4hperday.Suchtreatmentwithdinitro- tively instantaneous, whereasa morphological measure phenol results in "loose-coupling" of oxidative phos- iscumulative. Hence, evenasmall physiological differ- phorylation,oxidation ofNADH,anddiminishedATP encecan, overtime, producea largedifference in mor- Ill II-ROCHRONY DOSE-RESPONSE IN HYDROIDS 53 phology. The same logicsuggeststhat thissort ofdose- ofseavvater (filtered to 0.2/jm). Chamber temperature responserelationshipcanbestbeevaluatedbymeasures washeldconstant(20.5 0.02C)bymeansofanexter- thatareoffsetintime.Inotherwords,themorphological nal circulationwaterbath(Neslab RTE-100D),andthe effect of a physiological dose measured at one time rate ofdecline in oxygen concentration over a 30-min shouldbeassessed by morphological measurestaken at period was measured (using a Strathkelvin 1302 elec- latertimes.Giventheseconsiderations,themeanofnine trode and 781 oxygen meter) with stirring (by slowly measures (from threecycles ofthree stolons) ofgastro- spinning the magnet, cover slips, and colony). Subse- vascularflowtakenfromeachcolonyatthetimethesur- quently,thechamberwasopened.0.021 ml ofseawater facebecamecoveredwasusedtopredict morphological removed, 0.021 ml of 1 mAIdinitrophenol solution in outcomes taken subsequently (i.e., at the initiation of seawater added, the solution mixed and aerated thor- medusaproductionandatthefirstreleaseofmedusae). oughly with a small pipette, and the chamber resealed Dose-responserelationshipswereassessedgraphically (thisproceduretook ~7 min).Therateofdeclineinox- andbyusingparametricandnonparametricmeasuresof ygen concentration was then measured for another correlation.Gastrovascularflowrate(i.e.. lumenampli- 30min. Experiments were performed 3-5 h after the tudedividedbycycleperiodandstolonwidth)wasused subject colony was fed, as part ofthe normal feeding asthepredictor,andtheratiooftotal polypareatototal schedule. For the five colonies, the entire experiment unencrusted inner area was used as the morphological thustooknearly2weeks. outcome(theresultswerefoundtobe insensitivetothe Aftereachcolonywastested,acontrolexperimentwas particular morphological metric used). Both measures carriedoutwith thesamefivecoloniesandan identical werenaturallogtransformedtobettermeettheassump- protocol,exceptthatinsteadofaddinguncoupler.plain tionsofthe parametric correlations. However, particu- seawaterwasadded priorto the second measure ofthe larly in thecaseoftheoverfeedingexperiments, theas- declineinoxygenconcentration.Followingthesecontrol sumption ofa bivariate normal distribution is notwell experiments,feedingexperimentsweredone,againwith satisfied.Althoughall measuresofcorrelationweresim- thesame fivecolonieson the normal feedingschedule. ilar,inthiscasethenonparametriccorrelationsaremost Inthiscase,asimilarprotocolwasused,exceptthatafter appropriate, and only Spearman's Rsare reported here the first 30-min measure ofthedecline in oxygen con- (Sokal and Rohlf. 1981). Correlations between individ- centration, thecolony wasremoved from thechamber, ualflowparameters(lumenamplitude,cycleperiod,and fed a small amount ofbrine shrimp, carefully cleaned stolonwidth)andthemorphologicaloutcomewerealso ofanyshrimporshrimpportionsremainingoutsidethe tested. gastrovascular cavity, and returned to the chamber, Finally,itshouldbenotedthatthedose-responserela- whichwasthenresealed(thisproceduretook ~20min). tionships for the feeding manipulation and the uncou- Thedeclineinoxygenconcentrationwasthenmeasured pling experiments may not be directly comparable. In again. Ineach oftheseexperiments,datawereanalyzed particular, previousworkershaveshown thatconfining usingapaired-comparison/test. colonies in petri dishes can lead to more sheet-like growth(Miillerelat.. 1987; Plickert el a/.. 1987; Lange .Venture*ofmitochondrialredoxstale and Miiller, 1991). In such confined cultures, soluble morphogeneticfactorsproducedbythecoloniescanac- SpectrofluorometricassaysofNADVNADH provide cumulateandenhancepolypandstolonproduction.The a useful measure of mitochondria! redox state (e.g., dinitrophenolexperimentsincludethiseffectinallthree Chance and Baltscheffsky, 1958; Chance and Thorell, treatmentsand are thus not directly comparable to the 1959; Chance et til.. 1963; Chance. 1991). These mea- entirelyaquarium-growncoloniesofthefeedingmanip- suresare carried out //; vivo, with no apparent damage ulationexperiments. tothecolonies,usingaPerkin-Elmerspectrofluorometer (excitation at 366nm and emission read near460nm) Measuresofoxygenuptake andacryliccuvettes(whichdo notabsorboremitwhen excitedabove300nm).Similarexperimentswithavari- Fivenewlyexplantedclonalreplicateswereallowedto ety ofclonesofdifferent hydroid species(includingthe grow on 12-mm cover slips for 2weeks. At that point, P. carnea clone used here) have already demonstrated theeffectof30p.Mdinitrophenol on therateofoxygen that uncoupling with 30ftM dinitrophenol triggers a consumptionofeachcolonywasmeasured.First,acover shift of the redox state in the direction of oxidation slip,with itscolonyattached,wasaffixedwithadropof (BlackstoneandBuss, 1992, 1993;Blackstone,inpress). siliconegreasetoacoverslipcementedtoasmall mag- Here, the effects offeeding were investigated. Eighteen net.Thisassemblywascontained in a 13-mm diameter newly explanted clonal replicates were allowed to sealedglasschamber(Strathkelvin RC300)with 0.7 ml completely cover both sides of 12-mm cover slips 54 N. W. BLACKSTONE (~2 months).Toproduceanadequatesignal,threecol- oniesoncoverslipswerereadsimultaneously.Thusthe effectivesamplesizeis/;=6.Theemissionofeachgroup wasmeasured;thecolonieswerethenremovedfromthe cuvette, fed a small amount ofbrine shrimp, carefully cleanedofanyshrimporshrimpportionsremainingout- side the gastrovascular cavity, and returned to the cu- vette forasecond measurement ofemission. A control experiment was performed with the samecoloniesand an identical protocol, exceptthatcolonieswere not fed between emission readings. Data were analyzed using paired-comparison/tests. Results Feedingmanipulationexperiment* RepresentativecolonyimagescanbeseeninFigure 1 andontheWorldWideWeb(http://www.bios.niu.edu/ eande/blackstone/blackstone.html). Feeding manipula- tion substantially affects colony morphology such that higher rates offeeding produce colonies with a greater areaofpolypsand alesserunencrusted areawithin the colony(Fig. 3).Atthetimetheycompletelycoveredthe surface, coloniesin each treatment are roughly similar, but there is a small between-treatment effect (MANOVA, F = 2.93, df = 4, 28, P < 0.05). Subse- quentlyincolonydevelopment,thedifferencesbetween treatmentsbecome profound (Fig. 3; MANOVA at the Piniti0a.ti0o0n1o;fMmAeNduOsVaAdevateltohpemfeinrstt.reFle=ase13o.f2.mdefdu=sa4,e,28F, = 24.7, df= 4, 28, P 0.001). At thetimeofcovering thesurface,thereisalsoasignificantbetween-treatment differenceinflowrate(Fig.4;usingthecolonies-within- treatmenteffectastheerrorterm,F= 8.9,df= 2, 15,P <0.01)suchthatahigherrateoffeedingdiminishesthe flow. Since flow rate is a composite ofthree measured flow parameters (lumen amplitude, cycle period, and stolonwidth),itisilluminatingtoexaminethebetween- treatmentdifferencesinthesevariablesindividually.Lu- men amplitude and cycle period show no significant differences(Fig.5;F=0.20.df= 2, 15,P>0.8,andF= 1.43,df=2. 15. P> 0.25, respectivelyforeachvariable using the colonies-within-treatment effect as the error term). Stolon width, however, shows a dramatic be- tween-treatmentdifference(Fig. 5:F= 23.8,df= 2, 15, P <? 0.001, again using the colonies-within-treatment effect as the error term). A higher rate offeeding pro- ducesthickerstolons,andthebetween-treatmentdiffer- ences in flow rate derive not from differences in the amountofflow(amplitudeandperiod),butfromdiffer- encesinthethicknessofthestolons. Uncouplingexperiments RepresentativecolonyimagescanbeseeninFig. 1 and ontheWorldWideWeb(attheURLgivenintheprevi- HETEROCHRONY DOSE-RESPONSE IN HYDROIIXS 55 5- 56 N. W. BLACKSTONF. 140' B amplitude(um) 120: Dstolonwidth(um) 100: C] cycleperiod(s) 80-j 60: 40: 20- 23456 I I I I *c roo oo coo oo PARAMETERS o Sx* FLOW 140- 120: TOO: 60-j 40-j 20-
