Courtship, Copulation, and Sperm Transfer in Leucauge mariana (Araneae, Tetragnathidae) with Implications for Higher Classification PDF
Preview Courtship, Copulation, and Sperm Transfer in Leucauge mariana (Araneae, Tetragnathidae) with Implications for Higher Classification
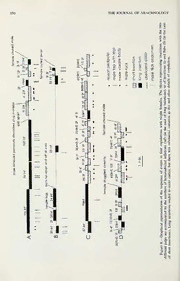
1998. The Journal of Arachnology 26:342-368 COURTSHIP, COPULATION, AND SPERM TRANSFER IN LEUCAUGE MARIANA (ARANEAE, TETRAGNATHIDAE) WITH IMPLICATIONS FOR HIGHER CLASSIFICATION William G. Eberhard: Smithsonian Tropical Research Institute, and Escuela de Biologia, Universidad de Costa Rica, Ciudad Universitaria, Costa Rica Bernhard A. Huber Escuela de Biologia, Universidad de Costa Rica, Ciudad Universitaria, Costa Rica ABSTRACT. ThecourtshipbehaviorofmaleLeucaugemariana (Keyserling 1881) spidersthatoccurred both prior to and during copulation is described, along with the positions and movements of the male genitalia. The great variation in male behavior suggests that it does not function in species recognition. Several kinds of female response are necessary if a male is to successfully inseminate her. Males made two types of insertion, involving different movements of palpal sclerites, and copulations with virgin females differedquantitativelyandqualitativelyfromthosewithnon-virgins. Malesdepositedencapsulated sperm and othermaterial in an outerchamberofthe female's spermathecaearly incopulation. Laterstages of copulation involved deposition of material on the surface of the female's epigynum that sometimes resulted, with the apparent addition ofmaterial by the female, in the formation ofaplug onthe epigynum. Sperm were decapsulated in the female soon after insemination, perhaps as a result of the action of a female glandular product, and later were found in two other chambers of her spermathecae. Contrary to previous discussions, male and female cheliceral clasping behavior accompanying copulation does not explain why the palpal morphology of these spiders is relatively simple, Cheliceral clasping was similar, though not identical, to that of several other tetragnathine spiders. Cheliceral clasping and details ofhow male palps engage the female may provide synapomorphies linking Leucauge to tetragnathines. RESUMEN. Se describe el comportamiento de cortejo de machos de Leucauge mariana (Keyserling 1881) que ocurrio antes y durante la copula, y las posiciones y los movimientos de la genitaliadelmacho. La gran variacion en el comportamiento de los machos sugiere que esto no funciona como senal de reconocimiento de la especie del macho. Varias respuestas de las hembras son necesarias par que un macho logre inseminarla exitosamente. Los machos efectuan dos tipos de insercion de los palps en los cuales realizaron diferentes movimientos con los escleritos del palpo. Las copulas con hembras virgenes diferieron tanto cuantitativamente como cualitativamente de las copulas con hembras no virgenes. Los machos introdujeron espermatozoides encapsulados y otras sustancias en una camera de la espermateca de la hembra durante una etapa temprana en la copula. Despues depositaron materiales sobre la superficie del epigeno que a veces formaron, en combinacion con material proveniente de lahembra, untapon sobre el epigeno. Los espermatozoides salieron de las capsulas dentro de la hembra, quizas como resultado de la acciondeunproducto glandulardelahembra, y despues llegaron adosotras camerasdelaespermateca. A1 contrario de algunas discusiones previas, el agarre entre los queliceros del macho y la hembra no explica porque la morfologia de los pedipalpos del macho de este grapo es relativamente sencilla. El agarre entre queliceros se asemeja al agarre de varias otras especies de Tetragnathinae, y este comporta- miento, mas otros detalles de como los palpos del macho se acoplan con la genitaliade lahembra, pueden proveer sinapomorfias que ligan Leucauge a Tetragnathinae. Male courtship behavior is often thought to male to initiate copulation. Ifthis is true, then function to induce the female to allow the male courtship behavior that occurs after cop^ ulation has begun (“copulatory courtship”) is ' Current address: Dept, of Entomology, American seemingly functionless and thus paradoxical. Museum of Natural History, Central Park West at It appears, nevertheless, that copulatory court- 79th St., New York, New York 10024 USA. ship is widespread in insects and spiders 342 EBERHARD & HUBER—COURTSHIP AND COPULATION IN LEUGAUGE 343 (Eberhard 1991, 1994; Huber in press). It iana (Keyserling 1881) to determine the pos- seems likely that copulatory courtship serves sible significance ofmale copulation behavior to induce other post-intromission female re- that may be linked to events inside the female sponses that are also critical to male repro- during copulation. We also describe the mor- ductive success, such as allowing the copula- phological mesh between male and female tion to go to completion, sperm transport genitalia, movements of the male genitalia, (Bukowski & Christenson 1997), dumping the the process of sperm transfer, and the phylo- sperm of previous males, storing and main- genetic implications of some aspects of Leu- taining the sperm of the current male, ovipo- cauge sexual behavior. sition, and refusing the sexual advances of METHODS other males (see Eberhard 1996 for a list of 20 possible female responses). Spiders were observed both in the field and There is a sizeable, though somewhat scat- in captivity during September and October tered, literature on spider mating behavior (re- 1989 and November 1995 near San Antonio viewed by Robinson 1982; Jackson & Macnab de Escazu, and February-November 1995 in 1991; Richman & Jackson 1992; and Huber in San Pedro de Montes de Oca (both in SanJose press; formorerecentworkon araneoidsLubin Province), Costa Rica. More than 40 pairs 1986; Gonzalez 1989; Gonzalez & Armendano were observed courting and mating in captiv- 1995; Castro 1995; Bukowski & Christenson ity using a 8X, 20X, 40X, and 80X dissecting 1997). Although there are descriptions ofmale microscope; verbal accounts of some copula- & courtship during copulation (e.g., Jackson tions were taped. Ten copulations were video- Whitehouse 1989 on the salticid Thorellia en- taped in captivity at 30 frames per second us- sifera [= Thorelliola ensifera (Thorell 1877)], ing a National Newvicon VHS camera in which male tapping appears to induce the equipped with +6 closeup lenses. One mating female to remain quiet; see also Stratton et al. sequence was videotaped in the field. All 1996; and Huber in press summarizing the ex- drawings depicting the behavior of entire an- tensive observations by U. Gerhardt), the em- imals were traced fromvideo images. Portions phasis has generally been on pre-copulatory of the spiders that were not resolved in the courtship. There are several reasons, however, videos were not drawn. Females whose mat- to suspectthatreports have beenbiased against ing history was known were obtained by col- descriptions of courtship behavior after copu- lecting penultimatejuvenile females that were lation has begun (Eberhard 1994). accompanied by adult males, allowing the fe- The sexual biology of spiders in the large males to molt to maturity in captivity (in all (> 100 species) genus Leucauge White 1841 cases this occurred within three days or less), has been little studied. Both newly molted vir- and then mating them one to seven days later. gin females and older females mate in the The silk lines on which the spiders met var- field (Eberhard et al. 1993). Males in the field ied, and seemed to have no effect on subse- tend to associate with immature females about quent courtship and mating. The female was to molt to maturity rather than with mature allowed to spin a few lines in an empty wood- females, suggesting that sperm from a fe- en or plastic rectangular frame at least 30 cm male’s first mating are more likely to fertilize on a side, orto rest on the orb ofanother adult her eggs (Eberhard et al. 1993). Castro (1995) female. All males observed in captivity had described several aspects of the pre-copula- been collected less than three hours previous- tory courtship in L. venusta (Walckenaer ly; no male was observed more than once. 1841), L. “mandibulata” (the specimens were Each pair’s behavior was followed until one of mariana - H.W. Levi pers. comm.) and the of the spiders decamped, or until neither had closely related Plesiometa argyra (Walcken- moved for at least 15 min. aer 1841). Brief descriptions of copulatory The palps of males frozen in liquid N dur- 2 courtship behavior in L. mariana and three ing copulation failed to remain coupled to fe- other, unidentified species of Leucauge were males. The positions ofpalp structures during given by Eberhard (1994). Female L. mariana hematodochal expansion were therefore deter- build egg sacs on the ground, and cover them mined by squeezing the pedipalp of a copu- with particles of soil and debris (Ibarra et al. lating male nearthe base with apairofforceps 1991). Here we use the very abundantL. mar- during maximal hematodochal expansion, cut- — 344 THE JOURNAL OF ARACHNOLOGY ting the connection to the male, and then plunging the still inflated pedipalp into Du- boscq-Brasil fixative (Romeis 1989). AL though the angle ofthe cymbium with the tib- ia straightened, the hematodochae remained fully inflated, and the positions of the bulbal sclerites were unchanged after the palp was fixed. The internal anatomy of male and female genitalia was determined using serial semi- — thin sections (IfjLm) of specimens fixed in Figure 1. The forward movement of rocking ethyl alcohol or Duboscq-Brasil, then embed- behavior by a courting male in lateral view (dotted ded in ERL-4206 epoxy resin and stainedwith lines follow solid lines by 0.07 sec). The male methylene blue in an aqueous borax solution rocked his body forward and backward by alter- (1%) (see Huber 1993). Voucher specimens nately extending and flexing his legs IV. have been deposited in the Museum of Com- parative Zoology at Harvard University, the American Museum of Natural History, and intact orbs, and on special molting webs Museo de Zoologia of the Universidad de (Eberhard et al. 1993). Of the behavior pat- Costa Rica. terns that males performed before copulation, at least seven may function to stimulate fe- RESULTS males (all were usually performed while the Pre-insertion courtship by males. The male was on the same line on which the fe- term “courtship” is used here to refer to be- male was resting): havior that was repeated both within and be- 1. Jerk: The male, while facing the female, tween pairs, that obviously resulted in stimuli flexed his anterior legs strongly and quickly being received by the other spider, and that (less than 0.1 sec) without releasing the lines had no obvious mechanical function in bring- they held. The result was a sharp jerk on the ing and keeping the spiders together (i.e., web that caused the female’s body to swing. walking toward the female is excluded). The These jerks were superficially similar tojerks term “copulation” is used to include all gen- spiders made in response to prey or other spi- italic contact between a particular male-fe- ders on their webs, and may represent search- male pair until the male left or became im- ing behavior rather than courtship. This be- mobile. The term “insertion” designates the havior was similar to that described as “jerk” entrance of the embolus and conductor into or “shake” in the courtship of a variety of the epigynal opening. araneids and nephilines by Robinson & Rob- Pre-copulatory courtship was both complex inson (1980), and the “jalon” of Castro and highly variable, and the descriptions be- (1995). low are only a preliminary list ofthe different 2. Rocking: The male flexed and extended types of behavior. The more complex ques- his legs IV rhythmically so that his body tions offrequencies and sequences ofdifferent rocked backward and up, then forward and behaviors are mostly ignored. The substantial down (Fig. 1). In several males vigorous variation in the simple presence or absence of bursts ofrocking were accompanied by small- particular types of behavior (e.g., Figs. 3, 8) er, more rapid flexes that set the male’s entire suggests that frequencies, durations, and se- body quivering briefly. Rocking movements quences of different behavior patterns also were often performed while the male faced may be quite variable. The names correpond, away from the female, but also occasionally when possible, to names used by Robinson & while he faced her. This behavior may corre- Robinson (1980) in their review of araneid spond to “vibracion o bamboleo” of Castro and nephiline courtship behavior. We have il- (1995). The most similar behavior described lustrated many behavior patterns due to the by Robinson & Robinson (1980) is the difficulty we experienced in comparing our “bounce”, but this is apparently an up-and- observations with those in previous accounts. down rather than a forward-and-backward Courtship and copulation occur in nature on movement as in L. mariana. EBERHARD & HUBER—COURTSHIP AND COPULATION IN LEUCAUGE 345 — Figure 2. Abdomen bobbing during pre-copu- latory courtship (dotted lines follow solid lines by — 0.1 sec) in ventral view. The male’s abdomen was Figure 3. Ventral view of rapidly alternating repeatedly flicked dorsally briefly. palp rubbing movements during pre-copulatory courtship. Theposteriormovementofthepalp (dot- ted lines on left of drawing, which follow solid 3. Abdomen bobbing: Abdomen bobbing lines by 0.07 sec) was followed 0.07 sec later by consisted of quick, dorsally directed flexions an anteriormovementofthe otherpalp (dottedlines ofthe male’s abdomen at the pedicel that last- on right follow solid lines there by 0.1 sec). ed about 0.07 sec each (Fig. 2). On some oc- casions it appeared that the abdomen vibrated porting the possibility that femur-chelicera as it was twitched, while on others a mule contact was an important aspect of these flicked his abdomen without causing a general movements. Inspection of a male’s cuticle vibration ofhis body, suggesting thatthese are with a compound microscope failed, however, two different movements. One common con- to reveal any special structure where the pal- text for abdomen bobbing was at the end of a pal femur contacted the chelicerae. burst ofpalp vibration (e.g.. Fig. 4). Abdomen Palp rubbing movements were termed “os- bobbing occurred both when the male was cilacion de palpos” by Castro (1995). They facing toward and away from the female. Ab- appear to be similar to the “palpal scrab- domen bobbing was similar and possibly ho- bling” described by Robinson & Robinson mologous to rapid “abdomen wagging” (1980), but differ in being performed while movements that occur in a variety of araneid the male was not in contact with the female. spiders (Robinson & Robinson 1980), and the Palp rubbing movements were much more theridiid Latrodectus (Ross & Smith 1979). rapid than those of palp cleaning when the 4. Palp rubbing: The male moved his ped- male passed his palps through his mouthparts ipalps in briefbursts of alternate anterodorso- following copulation. posteroventral movements, with the bulb 5. Twanging: The male folded his legs III moving from in front of his chelicerae to just ventrally and strummed the line under which ventral to his endites (Fig. 3). Bursts lasted up he was walking or hanging repeatedly with to several seconds (Fig. 4), andthe palps com- alternating lateral movements of the two legs pleted a single rub on the order of about one (Fig. 5). Twanging always involved a series of every 0.2-0.3 sec (Fig. 3); in some cases palp strums, and seemed particularly common at movements became progressively more brisk close range, during the final approach to the toward the end of each burst of vibration. female prior to cheliceral clasping (Fig. 5). Observations at 20X showed that the palps This behavior occurs in many araneids (Ger- & themselves did not usually touch each other hardt 1928; Robinson Robinson 1980; also during rubbing; in most cases the bristles on Blanke 1973, 1986 on Araneus cucurbitanus each cymbium probably contacted each other, [= Araneus cucurbitinus Clerck 1757]; Berry but this was sometimes not the case. The base 1987 on Cyrtophora moluccensis (Doleschall of each palpal femur rubbed against the retro- 1857)). It was noted by Castro (1995) only in lateral surface of the chelicera during palpal Plesiometa argyra. rubbing, and in some cases one palp was 6. Line tapping: The male rested under a moved while the other was immobile, sup- line leading toward the female, holding it with ^ 346 THE JOURNAL OF ARACHNOLOGY A P iiniiMiiiiiii liii III mil IIII i^ Li u u^ II II III III III ILII R 1 lll!ii!!:!!lllll!ll!lllllllllHillIlia 11 A 1 II 1 1 1 1 1 ! 1 1 II P ^liU^ JIMIIJIL _1LJ lUlllllllllll II llllll III III Hill i III 11 1II iL_ 5 sec RJML ILJIH illlllllill A 1 _ _L _ 1 P ML III! III! III! Ill IIIJ III! III! III! III! Ill Hill II ill III Hill III! Ill IB nil II III Rjimiiij QliUMJ Illlllllill — Figure 4. Patterns ofoccurrence ofabdomen bobbing (A), palp rubbing (P), and rocking (R) in three pre-copulatory courtship sequences in video tapes ofone male courting a virgin female. Each vertical line represents a burst ofmovements. Bursts ofrocking and palp rubbing tended to occurin groups. Abdomen bobbing tended to occur in conjunction with palp rubbing, while rocking tended to occur alone. his partially flexed legs II. Legs I and/or II touch or tap her briefly with his anterior legs, were held near the line and made quick, probably with the tarsi or metatarsi. Often af- mesally directed taps against the line (Fig. 6). ter such contact a male turned and moved The legs apparently did not grasp the line at away several body lengths, then attached his any time during slapping movements in which dragline and returned to her along it. Tapping the tarsus or metatarsus contacted the line. behavior did not seem to be stereotyped with Usually there were several taps in each series respect to either the parts ofthe female’s body (e.g., Fig. 6). This movement appears not to that were contacted or the pattern of move- have been described previously, at least in ments of the male. This behavior might thus these terms. be considered searching or sensory behavior 7. Tapping thefemale: The male, especially ofthe male ratherthan courtship. Nevertheless when interacting with a relatively non-aggres- in some pairs it was the only apparently stim- sive female, often approached close enough to ulatory behavior performed by the male be- — Figure 5. Twanging with one leg III by a courting male (stippled) seen in ventro-lateral view as he approached a female whose chelicerae were already open to clasp his (dotted lines follow solid lines by 0.07 sec). The male used alternate strokes with his legs III to strum the line along which he was moving toward the female. EBERHARD & HUBER—COURTSHIP AND COPULATION IN LEUGAUGE 347 / sec 1 Figure 6.—Line tapping during pre-^copulatory courtship by a male seen in ventro^-lateral view (dotted lines follow solid lines by 0.1 sec). The male's right leg 11 moved mesally to apparently tap the line on which he was resting, and immediately moved laterally. His right leg I also moved mesally, hitting the line slightly later than leg II. In the graph each vertical bar is a tapping movement. fore the female assumed the receptive posture peatedly openedand closedhercheliceraepri- and copulated. or to linking with the male (e.g.. Fig. 5); pre- Female responses.—We did not attempt to sumably these were intention or exploratory associate particular types of female response movements associated with cheliceral clasp- with specific male behavior patterns (in most ing. video recordings of male behavior the female 3. Assume mating posture: Just prior to was not in view); the general impression was copulation, the female loweredherbody while that there was little ifany specificity infemale facing directly toward the male, spreading her responses to particular male signals. Females anterior legs and opening her chelicerae wide, made three types of responses to male court- and often flexing her abdomen ventrally in an ship preceding copulation. acceptance posture (Fig. 5). The female clear- 1. Turn toward male: The female usually ly benther abdomen ventrally in 11 of 12 vid- turned to face the male when he approached eotaped pairings in which the angle of view- herfrom the rear, sometimes however only af- ing was adequate to resolve this detail. In two ter the male performed repeated bouts of cases the female later bent her abdomen dor- courtship behavior. sally while the male was attempting to insert 2. Open chelicerae: The female often re- his palp, and in one of these pairs he was un- 348 THE JOURNAL OF ARACHNOLOGY — Figure 7. Frontal view ofthe chelicerae ofmale and female L. mariana drawn to the same scale. The anterior surface of the basal segment ofthe male chelicerae has more setae, and a “ledge” that contacts the basal segment ofthe female chelicerae (perhaps the distal tooth) while she clasps his chelicerae with hers. able to reach her epigynum as a result. The licerae. Unfortunately the abundant hairs on male often tapped the female with his legs as the border of the female chelicerae made it she lowered herself into position and as she impossible to see the exact position of the fe- waited there. — male’s tooth with respect to the male’s ledge. Cheliceral clasp. Relatively stereotyped As the cheliceral clasp was being achieved or contact involving both the legs and the che- just after, the male extended one of his pedi- licerae of the male and the female occurred palps to rest on the ventral surface of the fe- just prior to copulation. The female always male’s abdomen. opened her chelicerae wide as the male ap- As the two spiders locked chelicerae, the proached (usually with his own chelicerae male positioned his legs I and II so that they closed), and then grasped the distal portions were in contact with the ventral surfaces of of the basal segments of the male’s chelicerae the corresponding legs of the female and by closing her fangs. The inner surface ofher tapped against them. Often his legs III were fang clearly pressed against the posterior sur- also held against the legs III of the female, face of the male’s chelicerae rather than contacting their dorsal surfaces. Usually the against his endites. The modihed “ledge” on male contacted the female with the distal por- the anterior surface ofeach ofthe male’s che- tions of his legs I and II (tarsi, metatarsi). licerae (Fig. 7) was thus pressed against the A given pair of spiders often made several distal surface of the basal segment of the fe- cheliceral clasps during a copulation (Fig. 8, male’s chelicerae. Observations at 8X with a Table 1). Between clasps the spiders moved mirror behind the spiders established that the apart, in some cases several body lengths. The female cheliceral tooth nearest the insertion of male often courted again before each subse- her fang was near the ledge on the male che- quent cheliceral clasp. In some cases the fe- EBERHARD & HUBER—COURTSHIP AND COPULATION IN LEUCAUGE 349 — Table 1. Characteristics (averages with one standard deviation) ofcopulations with virgin females and comparisons with copulations with females that had mated once 1-7 days earlier. Frequencies were com- pared using Chi Squared Tests; other comparisons were made using Mann-Whitney U Tests. Some cop- ulations were observed in more detail than others; this accounts for different sample sizes and missing data. ^Significantly different with Mann-Whitney UTest, P < 0.001. Female Virgin Female Mated {n = 24) {n = 13) P Duration copulation (min) 17.3 ± 6.1 9.9 ± 13.3 <0.001 Number long insertions 3.5 ± 2.0 0.2 ± 0.6 <0.001 Number bouts of 6.21 ± 5.2 4.1 ± 3.7 0.201 short insertions Number clasps with 2.3 ± 1.2 1.7 ± 1.4 0.032 chelicerae Female pushed palp with 50% 29% >0.1 leg III at least once Duration long insertions (sec) first 109 ±1\ {n = 24) second 123 ± 67 (« = 21) third 121 ± 104 {n = 14) Duration of each bout of short 40 ± 19 {n ^ 41 bouts, 7 copulations) insertions (sec) Number hematodochal expansions 57.0 ± 26.1* (/I = 34 insertions, 7 copulations) during each long insertion Number inflations during each 14.6 ± 7.0* {n -= 41 bouts, 7 copulations) bout of short insertions male’s behavior just after a pair broke apart legs tapped varied; frequently (butnot always) appeared to be aggressive, and she made rapid the right and left legs of a pair alternated. bursts of movement and gave relatively vio- Bursts oftapping usually lasted several sec- lent jerks on lines running toward the male. onds (average 4.5 ±1.2 sec, n = \3 bursts by The male nevertheless often courted and suc- one relatively actively tapping male). Leg tap- cessfully induced her to approach again (or ping occurred during the first moments after allow him to approach), and to assume the the female grasped the male chelicerae andthe acceptance posture. Copulations with virgin male attempted to insert his palp, and also females were longer, and included more chel- nearly always occurred during the withdrawal iceral clasps than copulations withnon-virgins of one palp and insertion of the other. When, (Table 1). — on occasion, there was a pause of a second or Copulation. 1. Leg and abdomen move- more between withdrawal and insertion, leg ments: During copulation males performed at tapping did not begin until several tenths of a least three behavior patterns seen in pre-in- second before the insertion occurred, suggest- sertion courtship: leg tapping with legs I and ing that insertion rather than withdrawal is the II, abdomen bobbing, and rocking. During leg context for leg tapping. Leg tapping also oc- tapping the male repeatedly tapped his ante- curred periodically during long insertions. The rior legs (I, II; sometimes also III) against the rhythm ofinflation and deflation ofthe male’s female’s legs, often on their ventral surfaces palpal hematodochae was not modified while (except for legs III). Tapping during copula- his legs tapped the female. tion differed from pre-copulatory tapping in Males also performed an additional behav- more consistently involving particular parts of ior not seen prior to insertion, bursts of front the female’s body. Each of the male’s legs leg pushing. The male’s front four legs were tapped on the corresponding leg ofthe female repeatedly extended synchronously againstthe (e.g., male right I on female left I, male right legs of the female while, in most cases, his II on female left II, etc.). The order in which legs III and IV were held immobile. Most ex- 1 350 THE JOURNAL OF ARACHNOLOGY two case the the (in with (f) 0 a ^O) -og" Eo insertions flubs D ^ -Q D and Q. -Q T) 1 D (i) 0CL tl u2 U copulation. D—0 0 representing 0 insertions O D D D of ^ E E E E ’•o ii of - blocks details or The other insertions), and females. this longin virginof case variation with the (in copulations substantial (inf) was four inflations in there events but of hematodochal earlier, sequenceof occur to the numbers of tended the by representation insertions accompanied Long Graphical u are — 8. palpsinsertions). Figure short different of EBERHARD & HUBER—COURTSHIP AND COPULATION IN LEUCAUGE 351 right I into the female’s epigynum. Male palps en- gaged—the female epigynum in two different ways “long” and “short” insertions. Long insertions usually occurred early in a copula- tion, and short insertions later, but there were numerous exceptions (e.g.. Fig. 8). In a long insertion, each palp usually made only a sin- gle long insertion before it was withdrawn from the female’s abdomen and the other was inserted (Fig. 8) (occasionally these distinc- tions were not clear, and the conductor and embolus withdrew from the female following each of the first few inflations of the basal hematodocha, and then re—mained inserted dur- ing subsequent inflations see descriptions of shortinsertions below, andFig. 8). In contrast, short insertions occurred in bursts of several short insertion attempts made by the same " ' palp before it was withdrawn and the other il li li II I !P 5illsec IS !! Ill II palp was extended to the female’s abdomen. — The duration ofa long insertion averaged over Figure 9. Rhythmic pushing with the front legs during copulation, seen with male (stippled) in an- a minute (Table 1), while short insertions last- tero-ventral view and the female in postero-lateral ed only on the order of a second or so. As view (dotted lines follow solid by 0.07 sec). While mentioned above, the first insertions in mat- the chelicerae were locked, the male’s legs I and II ings with virgin females were usually of the contacted the corresponding legs ofthe female and long type (Fig. 8), while copulations withnon- were extended and moved slightly forward (with virgins almost never included long insertions respecttothemale’sbody) insynchronywithpalpal (Table 1). The order of long and short inser- movements. Legs III and IV ofthe male were held tions was variable (Fig. 8); sometimes a long more or less still (female leg III was out offocus). insertion occurred after several short inser- The graph shows the pattern ofpushes by this male tions had been performed on the same side of (each vertical line is a push). The first push of a the epigynum. series was always stronger than the others. Both long and short insertions began in a similar manner. The palp was extended so that tension was at the male’s femur-patella joint the dorsal surface of the cymbium contacted (Fig. 9). Some males pushed only once each the ventral surface ofthe female abdomenjust time; more commonly, the male made repeat- anterior to her epigynum. The trochanter pro- ed quick series of pushes (Fig. 9). The jected ventrally, and the distal portion of the strength of the pushes varied widely. The tibia passed near the groove between the inner number ofbursts ofpushing during a long in- margins of the female coxae IV, but did not sertion ranged from 0-9, and averaged 2.4 for touch it (Figs. 9, 13). At least some of the each insertion that had at least one burst of many setae of the cymbium (Fig. 10), es- pushing (n = 8 copulations). Bursts of leg pecially those in its basal half, were interlaced pushing began at the same time as the basal among the setae near the female’s epigynum hematodocha of the palp was inflated. Defla- (Fig. 14). The cymbium was turned and di- tion, which was much more gradual than in- rected somewhat laterally (e.g., the male’s flation, occurred between bursts of pushes. right cymbium was directed to his left, so that 2. Movements of the male’s genitalia: In- its distal tip was just to the female’s right of sertion: During each cheliceral clasp, the male hermidline). Although it was difficultto make extended at least one palp one or more times direct observations, it appeared that the long to contact the female’s abdomen. During each patellar seta (Fig. 10) often (perhaps always) extension of a palp the basal hematodocha contacted the cymbium on its inner, concave was expanded one or more times to insert (or surface; in some cases this seta was displaced attempt to insert) the embolus and conductor laterally as the basal hematodocha was inflat-
