A taxonomic revision of the Meranoplus F. Smith of Madagascar (Hymenoptera: Formicidae: Myrmicinae) with keys to species and diagnosis of the males PDF
Preview A taxonomic revision of the Meranoplus F. Smith of Madagascar (Hymenoptera: Formicidae: Myrmicinae) with keys to species and diagnosis of the males
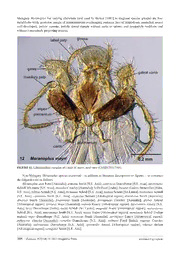
Zootaxa 3635 (4): 301–339 ISSN 1175-5326 (print edition) www.mapress.com/zootaxa/ Article ZOOTAXA Copyright © 2013 Magnolia Press ISSN1175-5334(online edition) http://dx.doi.org/10.11646/zootaxa.3635.4.1 http://zoobank.org/urn:lsid:zoobank.org:pub:9DB4A023-F9B4-4257-900B-F672F93CD845 A taxonomic revision of the Meranoplus F. Smith of Madagascar (Hymenoptera: Formicidae: Myrmicinae) with keys to species and diagnosis of the males BRENDON E. BOUDINOT & BRIAN L. FISHER 55 Music Concourse Drive, San Francisco, CA 94118, U.S.A. Corresponding author: [email protected] Table of contents ABSTRACT . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .301 INTRODUCTION. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .302 MATERIALS AND METHODS. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .302 Measurements . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .304 Indices. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .304 Repositories. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .306 SYNOPSIS OF MALAGASY SPECIES . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .306 DIAGNOSIS OF MALAGASY MERANOPLUS MALES. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .306 SPECIES GROUPS. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .307 Diagnosis of the M. mayri species group (worker). . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .311 Diagnosis of the M. nanus species group (worker). . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .312 KEY TO SPECIES . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .312 Key to workers . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .312 Key to queens . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .314 Key to males . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .316 SPECIES ACCOUNTS. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .316 Meranoplus cryptomysnew species. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .316 Meranoplus mayri Forel, 1910 . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .320 Meranoplus radamae Forel, 1891. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .327 Meranoplus sylvarius new species. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .333 ACKNOWLEDGMENTS. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .337 REFERENCES . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .338 ABSTRACT The species-level taxonomy of the ant genus Meranoplus F. Smith from Madagascar is revised. Two new species, M. cryptomys sp. n. and sylvarius sp. n. are described from workers and queens; M. mayri Forel, 1910, and M. radamae Forel, 1891, are redescribed, and queens and males for these two species are described for the first time. The first diagnosis of Meranoplus males for any biogeographic region is provided based on Malagasy species. Illustrated keys to all known Malagasy castes and species are presented. Diagnoses are given fortwo species groups: the M. mayri group and the M. nanus group. The diagnosis of the M. nanus species group from Bolton (1981) is thereby expanded with six new characters. Two species are known from the M. mayri species group and seven described species are known for the M. nanus species group, including the two new species described herein. The mouthparts, genitalia, and all castes, where known, of Malagasy Meranoplus are illustrated. Keywords: genitalia, gynes, Malagasy region, Meranoplini, species groups, wing venation Accepted by J. Longino: 5 Feb 2013; published: 28 Mar. 2013 301 INTRODUCTION Meranoplus is a unique and charismatic myrmicine genus of hairy, slow-moving, and armored ants. The genus is currently classified in its own tribe, the Meranoplini, with one fossil genus, Parameranoplus, from Baltic amber (44.1 ± 1.1 mya; Bolton 2003; Engel 2001; Wheeler 1915). The historic shuffling of Meranoplus through higher taxa—Cryptoceridae, Cataulacinae, Tetramoriini, Meranoplini—reflects our poor understanding of the phylogenetic position of Meranoplus within the Formicidae. Brady et al. (2006) recovered a clade of Meranoplus and Cataulacus, although this relationship was not supported in Moreau et al. (2006). The extant species of Meranoplus are distributed throughout the Old World, absent only from the Palearctic and Oceania regions but with the exception of M. levellei Emery, 1883, from New Caledonia (Fisher 2010; Wheeler 1935). The delineation of the genus Meranoplus is relatively straightforward; the genus is highly derived and unambiguously defined by several synapomorphies. The genus has been revised and reviewed across its entire distribution over the past few decades (Australasian region: Anderson 2006, Schödl 2004, 2007, Taylor 1990, 2006; Ethiopian region: Bolton 1981; and Oriental region: Schödl 1998, 1999). The genus is diagnosed by the compact mesosoma, which is dorsolaterally and often posterodorsally produced, and by the nine-segmented antenna with a three-segmented club (Bolton 2003). With over 80 valid species (Bolton 2012), it is predicted that over half of the Meranoplus diversityremains undescribed, most of these from Australia (Anderson 2006). Species of this genus are predominantly ground-nesting and, when disturbed, will display thanatosis enhanced by crypsis, i.e., individuals will accumulate dirt in their pilosity and play dead (Dornhaus & Powell 2010). With respect to diet, most species are omnivores and facultative granivores, while others, including the whole M. diversus species group, are specialist granivores (Anderson 2000, 2006). At least one species, the Malaysian rainforest-dwelling M. mucronatus F. Smith, 1857, is known to have a trophobiotic relationship with hemipterans (Maschwitz et al. 1987). Meranoplus species are known to be active both day and night (Gross et al. 1991), and to recruit via pheromone trails laid from the base of the sting using secretions from their extremely large Dufour glands (Hölldobler 1988; Billen et al. 2009; Billen & Taylor 1993). The function of the spatulate sting is still unknown (Kugler 1979). The only species of Meranoplus for which mating has been reported is M. peringuiyi Emery, 1886, in which mating swarms occurred after a rain and where males patrolled for the outnumbered females in a zig-zag manner (Robertson & Villet 1989; Schulmeister 2001). Male Meranoplus descriptions are few, varied, and scattered throughout the literature and across the biogeographic regions (Donisthorpe 1949; Forel 1915; Smith 1876). None of these descriptions provide diagnosis of Meranoplus males for any biogeographic region or locale, and most do not provide sufficient detail for genus- level identification. Given the potential of male ants to clarify the natural history of ant reproductive biology (Kaspari et al. 2001), improve the discovery and definition of genera (Yoshimura & Fisher 2009, 2011, 2012a), and aid ant taxonomy and systematics (Brady & Ward2005; LaPolla2004; Song & Bucheli 2010; Yoshimura & Fisher 2012b), including descriptions of male ants is a research priority. The biodiversity of Madagascar is exceptionally rich, with levels of endemism ranging from 65% for freshwater fish to 83–86% for non-marine plants, terrestrial vertebrates, and non-marine invertebrates (Goodman & Benstead 2005). Ants exhibit an endemism level of around 95% for the 1,300 Malagasy species, of which about 60% are undescribed (Fisher 2003, 2005; Hita Garcia & Fisher 2012). To this end, twenty-seven modern revisions in print or in press have contributed over 200 new species to the Malagasy ant fauna list. Here we present the first new species of Malagasy Meranoplus in over a century, with keys to all known castes, redescriptions of the workers of M. mayri Forel, 1910 and M. radamae Forel, 1891, the first descriptions of Meranoplus sexuals for Madagascar, and the first diagnosis of the male sex for the genus. MATERIALS AND METHODS Most specimens examined in this study were collected by Fisher et al. over the past two decades, provided with unique specimen identifiers affixed to each pin (viz. CASENT labels), and digitally databased. Every databased specimen record has been uploaded to AntWeb.org, a continuously updated resource that provides “living” distribution maps. Species distributions are illustrated in figs. 64–67. Anatomical terminology predominantly follows prior authorities: mouthparts (Gotwald 1969), wing venation (Yoshimura & Fisher 2012b), setational stature (Wilson 1955), and sculpture (Harris 1979). An exception to 302 · Zootaxa 3635 (4) © 2013 Magnolia Press BOUDINOT & FISHER Yoshimura & Fisher’s (2012b) venation terminology is the use of costal cell on the forewing, rather than costal+subcostal cell; no subcostal cell is present because of the fusion of the subcostal and radial veins in the Aculeata (Gauld & Bolton 1988). To summarize the sculptural terms: areolate is defined as raised ridges which connect to form polygons (cf. promesonotum in fig. 35); costate indicates linear, parallel raised ridges (cf. mesosomal pleurae in fig. 40); dense-punctate describes contiguous punctures (fig. 25); rugose describes non- linear raised ridges (fig. 51). Stellate punctures are defined as punctures having three or more radiating lines (fig. 26). In addition, many Meranoplus species have a carina on the ventral surface of the frontal carina above the scrobe, which is termed the scrobal carina (fig. 2). The term torular lobe is used as defined in Keller (2011), referring to the medial arch of the antennal socket torulus. The term promesonotal shield is unique to Meranoplus, and refers to the fused pro- and meso- segments of the mesosoma, which are often elongated as shelves laterally and posteriorly (Bolton 1981). Although abdominal segments IV–VII are distinctly constricted anteriorly in Meranoplus, we eschew the term gaster as it refers to different tagmata across the ant subfamilies (Keller 2011). For genitalia, we follow the terminology of Boudinot (2013) which reflects the homologies of male ant genitalia with basal Hymenoptera; terms utilized by Yoshimura & Fisher (2011) are indicated in parentheses. The genitalia are composed of the cupula (= basal ring) and three paired valves: the parameres, volsellae, and penisvalvae (=aedeagal plates). The lateral-most valve, the paramere, is composed of the basal basimere and distal telomere (= harpago). The volsella, medial to the paramere and lateral to the penisvalva in situ, and is composed of the basivolsella, cuspis, and digitus. The cuspis and digitus are the distal elements of the volsella, and can be recognized by their position and shape: the cuspis is lateral (closest to the paramere), lobe-like, and extends anteriorly (basally) as a setose plate, while the digitusis medial (near the penisvalva) and finger-like with the apex often directed ventrally. The penisvalva is divided into the valvura, the anterior (basal) apodeme,andvalviceps, the posterior (apical) broadened plate which often has teeth along the ventral margin. Specimen preparation for mouthparts and male genitalia was done as follows. To soften specimens, point- mounted or in > 90% ethanol, specimens were placed in 70% ethanol at least overnight. To dissect mouthparts, the labiomaxillary complex was pulled somewhat out of the headcapsule with a no. 3 entomology pin with the tip bent into a hook. Using forceps, the labiomaxillary complex was grasped from the base and pulled out of the head capsule, then mounted on a glass slide with a drop of KY Personal Lubricant (Johnson & Johnson Inc., Markham, Canada). The labrum was removed by gently grasping the base of the sclerite and pulling. To dissect the genitalia from softened males, the entire genital capsule was removed from the metasoma, leaving behind sternum IX and tergum X. Observations on the intact genital capsule were made. The genital capsule was then grasped by the forceps and cut in half using a pin, after which the right penisvalva was removed. This penisvalva and its associated paramere and volsella were then mounted as two units on the same slide. The left half of the genitalia was placed in a ~4 mm cup-shaped piece of cotton and rolled in a 1 cm2 piece of facial tissue. This was then inserted into a silicone-stoppered polyethylene microvial (BioQuip, Rancho Dominguez, California, U.S.A.) filled with 100% ethanol and placed upside-down in a vial of 100% ethanol with the voucher specimen. The slide mounts and voucher specimens were deposited in the CAS collection. Montage images of point-mounted specimens were generated using Leica Application Software Version 3.8 from micrographs captured via a Leica DFC 450 camera attached to a Leica Z16 APO dissecting microscope. All montages are available on AntWeb.org (http://www.AntWeb.org/). Compound microscope images were captured with a Leica DFC 500 camera mounted on a Leica MZ 16A microscope using LAS V. 2.5.0 R1 software; montages were generated with Helicon Focus 4.70 software. Measurements follow Schödl (2007) with additions and a few indicated acronym changes. Although of little value for delimiting Meranoplus species of Madagascar, measurements of the petiole and postpetiole are included for comparative purposes as they have previously been used for Meranoplus alpha taxonomy (Taylor 1996; Schödl 1998, 2007). Additionally, the scape index (SI) is conventionally calculated by dividing scape length by head length; we use a percentage representation of scape length divided by head length following Schödl (ibid.) to facilitate comparison to previous taxonomic works on the genus. Two systems were used to measure the spines to account for variation in the shape of the basal flanges of the propodeal spines. For workers, Longino’s (2003) measure of propodeal spine length is followed (cf. SPL below). For gynes, the basal inflection point at the base of spine was used, as the spines of this caste do not have appreciably variable profiles. Measurements were recorded in mm to three decimal places and were taken using two methods: with a digital Mitutoyo stage micrometer mounted on a Leica MZ 125 and an ocular cross-hair; or with an ocular micrometer THEMERANOPLUS OF MADAGASCAR Zootaxa 3635 (4) © 2013 Magnolia Press · 303 calibrated with a Leica standard (Art No. 10310345) which were either taken with a Leica MC165C or a Leica MC95. Only two significant figures are presented due to the use of the ocular micrometer, which reduced the precision of measurements. All measurements are illustrated (figs. 1–7). Measurements ATL: Abdominal Tergum IV Length. Maximum length of fourth abdominal tergum measured with anterior and posterior margins in same plane of focus. ATW: Abdominal Tergum IV Width. Maximum width of fourth abdominal tergum with anterior, posterior, and lateral borders in same plane of focus. CDD: Clypeal Denticle Distance. Distance between clypeal denticle apices, measured in full-face view. CW: Clypeus Width. Distance between the apices of the frontal lobes across the clypeus. EL: Eye Length. Maximum eye length in profile view. EW: Eye Width. Maximum eye width in profile view. HL: Head Length. Maximum length of head capsule, excluding mandibles, measured from anterior margin of clypeus to nuchal carina, with both in same plane of focus. HLA: Head Length, Anterior. Distance between the anterior edges of the eyes to the mandible bases in full-face view. HW: Head Width. Maximum width of head capsule behind the eyes, in full-face view. PML: Promesonotum Length. Maximum length of promesonotum from posterior spine/denticle apices to anterolateral denticle apices; all four apices in same plane of focus. (= PMD, Schödl 2007) PPH: Postpetiole Height. Measured from sternal process base to postpetiole apex in lateral view. PPL: Postpetiole Length. Measured from anterior to posterior inflections of postpetiole node in lateral view. PWA: Promesonotal Width, Anterior. Maximum width of promesonotal shield between anterolateral denticle apices in dorsal view. (= PW, Schödl 2007) PWP: Promesonotal Width, Posterior. Distance between posterior-most promesonotal spine or denticle apices. PTH: Petiole Height. Measured from petiole sternum to apex in lateral view. PTL: Petiole Length. Measured from anterior to posterior inflections of petiole node. SL: Scape Length. Maximum length of the scape excluding basal constriction. SPL: Propodeal Spine Length. Workers: distance from inner posterior margin of propodeal spiracle to propodeal spine apex. Gynes: maximum propodeal spine length from basal inflection of spine, to spine apex. WL: Weber’s Length. Maximum diagonal length of mesosoma from anterior inflection of pronotum to posterolateral corner of the metapleuron or the metapleural lobes, whichever is most distant. Indices CDI: Clypeal Denticle Index. CDD*100/CML CI: Cephalic Index. HW*100/HL CS: Cephalic Size. (HW+HL)/2 EYE: Eye Index. 100*(EL+EW)/CS OMI: Ocular-Mandibular Index. EL*100/HLA PMI: Promesonotum Index 1. PWA*100/PML (= PMI2, Schödl 2007) PPI: Postpetiole Index. PPL*100/PPH PTI: Petiole Index. PTL*100/PTH PWI: Promesonotum Index 2. PWP*100/PML SEI: Scape-Eye Index. EL*100/SL SI: Scape Index. SL*100/HW 304 · Zootaxa 3635 (4) © 2013 Magnolia Press BOUDINOT & FISHER FIGURES 1–7.Measurements of Meranoplus species. 1. Measurements with head in full face view. 2.Measurements of head in profile; scrobal carina indicated by “sc”.3. Scape length. 4. Mesosoma profile measurements. 5. Measurements of the petiole and postpetiole in profile. 6. Promesonotal shield measurements, dorsal view. 7. Abdominal tergum IV measurements, dorsal view. THEMERANOPLUS OF MADAGASCAR Zootaxa 3635 (4) © 2013 Magnolia Press · 305 Repositories Collections which provided material for study or to which types have been distributed are abbreviated as follows: BMNH The Natural History Museum, London, U.K. CASC California Academy of Sciences, San Francisco, California, U.S.A. MCZC Museum of Comparative Zoology, Harvard University, Cambridge, Massachusetts, U.S.A. MHNG Muséum d’Histoire Naturelle de la Ville de Genève, Geneva, Switzerland NHMB Naturhistorisches Museum, Basel, Switzerland USNM National Muséum of Natural History [Smithsonian], Washington, D.C., U.S.A. SYNOPSIS OF MALAGASY SPECIES Synopsis of Meranoplus species and species groups of Madagascar with known castes indicated in brackets (w, worker; q, queen; m, male): Meranoplus mayri species group mayri Forel, 1910. MADAGASACAR [w, q, m] Meranoplus nanus species group cryptomys sp. n. MADAGASCAR[w, q] radamae Forel, 1891. MADAGASCAR [w, q, m] sylvariussp. n. MADAGASCAR [w, q] DIAGNOSIS OF MALAGASY MERANOPLUS MALES Without a global or regional assessment of Myrmicine males, an indication of synapomorphies is not possible. For a diagnosis of the worker refer to Bolton (2003). 1. Labrum cleft medially (fig. 11). 2. Palpal formula 5,3 (fig. 12). 3. Cardo with egg-shaped lacuna (fig. 12). 4. Mandibles poorly developed, infrequently reaching midline of head when closed; spatulate to bladelike (figs. 31, 32, 48). 5. Antenna with 13 segments; antennomeres 5-13 swollen. 6. Scape short (SI < 33; figs. 31, 32, 48). 7. Eyes large (EL/HW 0.30-0.45; figs. 31, 32). 8. Ocelli present, about as large as antennal sockets (figs. 31, 32, 48). 9. Clypeus not produced posteriorly between antennal sockets (figs. 31, 32, 48). 10. Frontal lobes absent (figs. 31, 32, 48). 11. Frontal carinae distinct to absent (figs. 31, 32, 48). 12. Torular lobe present, raised. 13. Head with raised nuchal carina (figs. 46, 55). 14. Notaulus present (figs. 47, 56). 15. Parapsidal line present (figs. 47, 56). 16. Meso- and metatibial spurs present, single. 17. Forewing (FW) pterostigma present (fig. 17). 18. FW vein Rs+M splits distal to crossvein 1m-cu (fig. 17). 19. FW crossvein cu-a occurs about 2/3 along length of M+Cu (fig. 17). 20. FW crossvein 2rs-m absent (fig. 17). 21. Hindwing (HW): terminal segments of veins Sc+R1 and Rs with bases contiguous (fig. 18). 22. HW cell formed by the veins Sc+R, M+Cu and crossvein M+1rs-m narrow, apex acute (fig. 18). 23. HW cell formed by the veins 1A, M+Cu and crossvein cu-a less than 1/3 the length of the larger cell (fig. 18). 24. Petiole without pedicel (figs. 46, 55). 25. Postpetiole globose in lateral view; constricted posteriorly (figs. 46, 55). 26. Abdominal sternum XIII broadly emarginate. 306 · Zootaxa 3635 (4) © 2013 Magnolia Press BOUDINOT & FISHER 27. Abdominal sternum IX about as long as broad; narrowed apically. 28. Pygostyles present. 29. Telomere separated from basimere by a suture (figs. 13, 14). 30. Digitus long, fingerlike; apex curved or bent ventrally (figs. 13, 14). 31. Penisvalva subrectangular with serrate ventral margin (figs. 15, 16). Comments Comments correspond to character numbers as above. 4. Male mandibles vary, with some specimens having two denticles on the masticatory margin, some specimens having one den- ticle—variably in the basal or apical position—and some specimens edentate. Any combination of the above characters may be found differing between the left and right mandibles of the same specimen. 16. Meso- and metatibial spurs may be absent in the unknown male of Meranoplus sylvarius, for which the females lack these spurs. 25. The petiolar node varies much more than the postpetiole in development within populations, varying from subnodiform (poste- rior face somewhat distinct) to globose. FIGURES 8–11. Labrum of Meranoplus species, ectal view. 8. Meranoplus mayri worker (CASENT0317545). 9. Meranoplus radamae worker (CASENT0317561). 10. Meranoplus sylvarius worker (CASENT0317566). 11. Meranoplus mayri male (CASENT0317565). SPECIES GROUPS All four Malagasy species of Meranoplus are endemic and conform to Bolton’s (1981, 2003) diagnosis of the genus. Two additional shared traits potentially diagnostic for the genus but not mentioned in Bolton (2003) are the spatulate sting (Kugler 1978) and the presence of a denticle or strong swelling on the ventral side of the mandible beneath the third tooth from the masticatory margin apex. The Malagasy species may be split into the M. mayri and M. radamae groups with south Indian and Afrotropical affinities, respectively. Further characters shared by all THEMERANOPLUS OF MADAGASCAR Zootaxa 3635 (4) © 2013 Magnolia Press · 307 Malagasy Meranoplus but varying elsewhere (and used by Bolton [1981] to diagnose species groups) are four mandibular teeth; posterior margin of promesonotum overhanging posterior face of propodeum; propodeal spines well-developed; petiole cuneate; petiole dorsal margin without teeth or spines; and postpetiole nodiform and without a posteriorly projecting process. FIGURE 12. Labiomaxillary complex of a male M. mayri, ectal view (CASENT0317565). Non-Malagasy Meranoplus species examined—in addition to literature descriptions or figures— to construct the diagnoses are as follows: Meranoplus ajax Forel[Australia]; armatus Smith[S.E. Asia]; asteriscus Donisthorpe[S.E. Asia]; atronitudus Schödl MS-name [S.E. Asia]; aurealus Crawley [Australia]; bellii Forel [India]; bicolor (Guérin-Méneville) [India, S.E. Asia]; biliran Schödl[S.E. Asia]; birmanus Schödl[S.E. Asia]; boltoni Schödl[Sri Lanka]; borneensis Schödl [S.E. Asia]; castaneus Smith [S.E. Asia]; clypeatus Bernard [Afrotropical region]; dimidiatus Smith [Australia]; diversus Smith [Australia]; fenestratus Smith [Australia]; ferrugineus Crawley [Australia]; glaber Arnold [Afrotropical region]; hirsutus Mayr [Australia]; inermis Emery [Afrotropical region]; laeviventris Emery [S.E. Asia]; levis Donisthorpe [India]; loebli Schödl [Sri Lanka]; magrettii André [Afrotropical region]; malaysianus Schödl [S.E. Asia]; mucronatus Smith [S.E. Asia]; nanus André [Afrotropical region]; nepalensis Schödl [Indian region]; niger Donisthorpe [S.E. Asia]; oceanicus Smith [Australia]; peringueyi Emery [Afrotropical region]; pubescens (Smith) [Australia]; raripilis Donisthorpe [S.E. Asia]; rothneyi Forel [India]; rugosus Crawley [Australia]; sabronensis Donisthorpe [S.E. Asia]; spininodis Arnold [Afrotropical region]; sthenus Bolton [Afrotropical region]; vestigator Smith [S.E. Asia]. 308 · Zootaxa 3635 (4) © 2013 Magnolia Press BOUDINOT & FISHER FIGURES 13–14. Rightvolsella and paramere of Meranoplus species, in mesal view displaying the digitus, cuspis, basimere, and telomere. 13. Meranoplus mayri (CASENT0317564). 14. Meranoplus radamae (CASENT0115117). THEMERANOPLUS OF MADAGASCAR Zootaxa 3635 (4) © 2013 Magnolia Press · 309 FIGURES 15–16. Right penisvalva of Meranoplus species, ectal view. 15. Meranoplus mayri (CASENT0317564). 16. Meranoplus radamae (CASENT0115117). 310 · Zootaxa 3635 (4) © 2013 Magnolia Press BOUDINOT & FISHER
